Membrane Permeability Study Guide
Introduction
Cell membranes protect and organize cells. The structure and function of cells are critically reliant on membranes. The plasma membrane of a cell controls what enters the cell and how much of any given substance comes in.
Cell Membranes
Cell membranes protect and organize cells. The structure and function of cells are critically reliant on membranes. All cells have an outer plasma membrane that controls what enters the cell and how much of any given substance comes in.
The lipid bilayer has been determined as the universal basis for cell membrane structure with a common structural organization: bilayers of phospholipids with associated proteins.
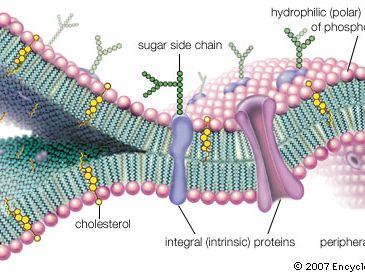
The principal components of a cell membrane are lipids (phospholipids and cholesterol), proteins, and carbohydrates. The amounts of proteins, lipids, and carbohydrates in the plasma membrane vary with cell type. Still, lipids constitute approximately 50% of the mass of most cell membranes, nearly all of the remainder being protein. Each component plays a role in the selective permeability of the membrane.
The Lipid Layer
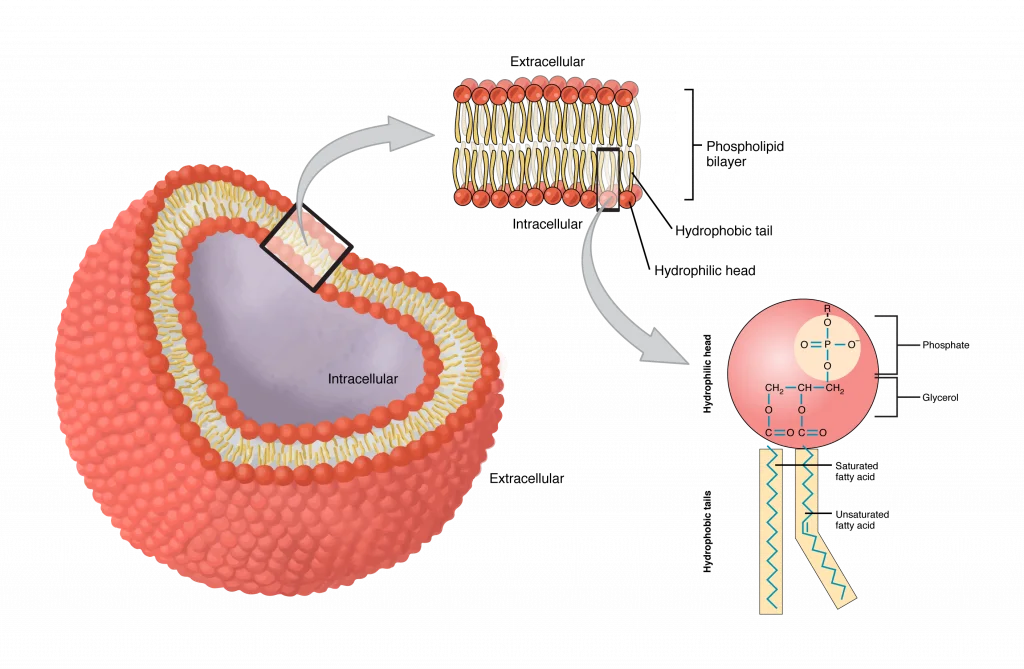
Biological membranes consist of a continuous double layer of lipid molecules in which membrane proteins are embedded. The main component of cellular membranes are lipids. There are three major classes of membrane lipid molecules—phospholipids, cholesterol, and glycolipids. Most cellular membranes are made of glycerophospholipids, molecules composed of glycerol, a three-carbon molecule that functions as the backbone, a phosphate group, and two fatty acid chains. Phospholipids are amphipathic molecules, as the two fatty acid chains are hydrophobic, linked to a phosphate-containing hydrophilic head group. When placed in water, the shape and amphipathic nature of the lipid molecules cause them to assemble spontaneously into bilayers.
The cell membrane consists of two adjoining layers of phospholipids. In water, these molecules spontaneously align with their negatively charged polar phosphate heads facing outward and their non-polar lipid tails lining up in the bilayer’s interior. Thus, the hydrophilic heads of the lipids in a cell’s plasma membrane face both the water-based cytoplasm Intracellular fluid (ICF) and the exterior fluid environment outside of the cell Extracellular fluid (ECF). This arrangement leads to the formation of a sealed compartment that separates the cell’s internal environment from the external environment.
The Cell Membrane as a Fluid Mosaic
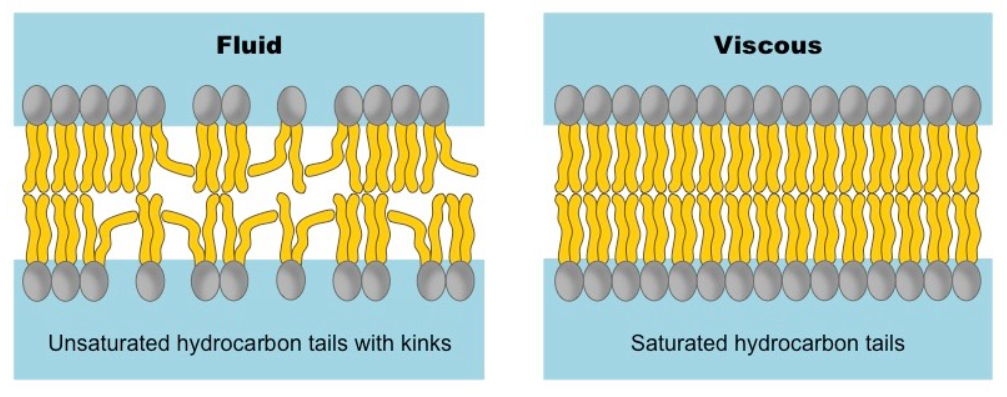
An essential property of lipid bilayers is that they behave as two-dimensional fluids with individual lipid and protein molecules that diffuse rapidly within their own monolayer. This means the lipids and proteins in the cell membrane are not rigidly locked in place as described by the fluid mosaic model. Membrane fluidity is determined both by lipid composition as well as temperature. Decreasing temperatures tend to cause the lipid molecules to pack together tightly, thus decreasing fluidity. Lipids comprising unsaturated fatty acids increase membrane fluidity because the presence of double bonds between adjacent carbon atoms introduces 30 degrees kinks in the fatty acid tails, making them more difficult to pack together. Thus, if saturated fatty acids with their straight tails owing to the absence of double bonds between adjacent carbon atoms, are compressed by decreasing temperatures, they press in on each other, making a dense and rigid membrane.
Eucaryotic plasma membranes contain substantial amounts of cholesterol that assist in maintaining fluidity. Cholesterol molecules insert into the bilayer with their polar hydroxyl groups close to the hydrophilic head groups of the phospholipids. This prevents lower temperatures from inhibiting fluidity and prevents increased temperatures from increasing fluidity too much.
Selective Permeability Is A Direct Result Of Membrane Structure
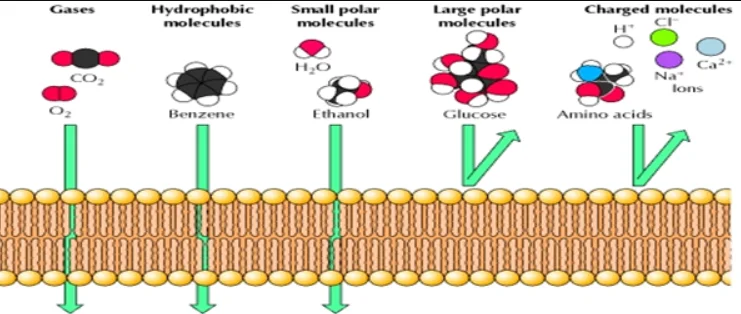
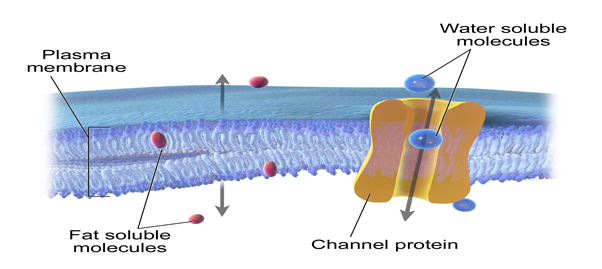
This structure of cell membranes results in selective permeability. The phospholipids are tightly packed together in the lipid bilayer, and the membrane has a hydrophobic interior due to the lipid tails. A membrane that is selectively permeable allows only substances meeting specific criteria to pass through it unassisted. In the case of cell membranes, only small uncharged molecules can diffuse freely through the lipid cell membrane. Small non-polar molecules, such as N2, O2, and CO2, are soluble in the lipid bilayer as the lipid tails of the membrane are non-polar and therefore can readily cross cell membranes. Thus, selective permeability of the cell membrane helps get rid of waste products, particularly carbon dioxide (CO2), which must leave the cell and bring in oxygen necessary for the cell during metabolism.
Small uncharged polar molecules, such as H2O, also can diffuse through membranes, but larger uncharged polar water-soluble molecules, such as glucose, amino acids, and electrolytes cannot. Charged molecules, such as Ca++, Na+, K+, and Cl–; and H+ ions cannot diffuse through a phospholipid bilayer regardless of size because they are repelled by the hydrophobic tails of the phospholipid bilayer.
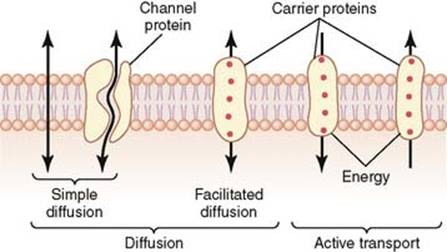
These molecules pass across membranes via the action of special transmembrane proteins, which act as transporters. Such transport proteins define the selective permeability of cell membranes. Proteins make up for roughly half the mass of most cellular membranes. Two different proteins commonly associated with the cell membrane are the integral proteins and peripheral proteins. Integral proteins contain multiple membrane-spanning regions that form a passage through the lipid bilayer. This allows polar or charged molecules to cross the membrane through a protein pore without coming in contact with the hydrophobic fatty acid chains of the membrane phospholipids. Membrane transport proteins are specific and selective for the molecules they move, and they often utilize ATP for this process.
There are two general classes of membrane transport proteins. Channel proteins form open pores through the membrane, allowing the free passage of any molecule of the appropriate size. Inorganic ions such as Na+, K+, Ca2+, and Cl- across the plasma membrane via ion channels. Once open, channel proteins form tiny pores through which ions of the appropriate size and charge can cross the membrane by diffusion. The pores are not permanently open; instead, they are selectively opened and closed in response to extracellular signals, allowing the cell to control the movement of ions across the membrane as observed in nerve and muscle cells. A channel protein is an example of an integral protein.
In contrast to channel proteins, carrier proteins selectively bind and transport specific small molecules, such as glucose. Once the carrier protein binds specific molecules it then undergoes conformational changes. This opens the channel and allows the molecule to be transported to pass across the cell and be released on the other side.
Conclusion
- The cell membrane is a selectively permeable gatekeeper between the cell and the extracellular environment.
- Its selective permeability due to the lipid bilayer makes certain that essential molecules such as glucose, amino acids, and lipids readily enter the cell, cell organelles remain in the cell, and waste compounds exit the cell.
- In short, the selective permeability of the cell membrane allows the cell to separate and maintain a constant internal environment from the external environment.
FAQs
1. Which factors affect membrane permeability?
The following factors affect membrane permeability:
- The medium composition
- Solute concentrations
- Temperature
2. How is phospholipid structure related to the selective permeability of the plasma membrane?
The phospholipid part of the plasma membrane renders the latter hydrophobic and therefore polar molecules would not be able to easily pass through this layer.
3. How does membrane permeability affect the rate of diffusion?
A membrane with smaller surface area or lower permeability will impede molecular movement and thus lead to slower diffusion.
4. How does phospholipid structure related to the selective permeability of the plasma membrane?
The structure of the plasma membrane makes it selectively permeable, enabling it to regulate the transport of substances into and out of the cell. For example, small, nonpolar molecules are hydrophobic, so they can easily cross the phospholipid bilayer of the plasma membrane. Polar molecules and ions are hydrophilic, so they cannot very easily cross the hydrophobic portion of the plasma membrane (formed by the phospholipid tails).
We hope you enjoyed studying this lesson and learned something cool about Membrane Permeability! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun VR classrooms – we promise, it makes studying much more fun 😎
Sources:
-
Rogers, Kara. “Cell Membrane | Definition, Function, & Structure.” Encyclopædia Britannica, 6 June 2019, www.britannica.com/science/cell-membrane.
-
“Membrane Fluidity | BioNinja.” Ib.bioninja.com.au, ib.bioninja.com.au/standard-level/topic-1-cell-biology/13-membrane-structure/membrane-fluidity.html.