Proteins Study Guide
INTRODUCTION
Proteins are the building blocks of our bodies. We classify them based on their structure, which is formed when amino acids condense together to form peptide bonds. The primary structure of every protein is a sequence of amino acids called a polypeptide chain. Proteins have different dihedral angles of their peptide bonds, which determine their secondary structure. The tertiary structure of a protein results from the way the protein chains fold in space. Lastly, quaternary structure is how folded polypeptide molecules form complex functional proteins. This is an overview of how protein is made. In this article, we will learn more about different protein structures.
PRIMARY PROTEIN STRUCTURE
Let’s take a look at some of the properties of the primary structure of a protein:
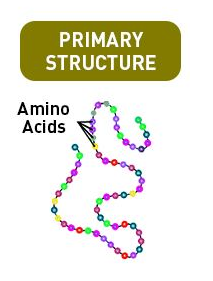
- The exact order in which amino acids are arranged to form chains is known as the primary structure of a protein.
- The final fold of the protein is determined by this sequence, which in turn determines the specific function of the protein.
- When polypeptides are chained together, they form proteins. The amino acids in these chains are arranged in a particular sequence. This sequence is the identity of the protein and the task it performs. Any disruption in the sequence can change the functioning of the protein.
- Genetic disorders like sickle cell anemia, albinism, etc., can be linked to alterations in primary protein structures.
SECONDARY PROTEIN STRUCTURE
Let’s look at some of the properties of secondary protein structures:
- Polypeptides are protein molecules that are chained together to form proteins, but they do not exist in a simple chain structure.
- Polypeptide chains tend to fold because of the interaction that happens between the carboxyl and amino groups of the peptide link.
- The secondary protein structure is the shape that long polypeptide chains form.
- The two popular structures formed due to the folding of chains are helix and pleated sheet structures.
TERTIARY PROTEIN STRUCTURE
- When the secondary protein structure folds further, it forms a tertiary structure.
- A number of forces are involved in stabilizing the tertiary structure.
- The two most commonly found tertiary protein structures are fibrous and globular.
- The electrostatic force of attraction is one of the major forces that stabilize the secondary and tertiary structures.
QUATERNARY PROTEIN STRUCTURE
- The manner in which tertiary structures are spatially arranged gives rise to quaternary protein structure.
- When a protein is made of two or more polypeptide chains, each chain is referred to as a subunit. The spatial arrangement of these subunits with respect to each other is a quaternary protein structure.
CONCLUSION:
- Protein structure is formed when amino acids condense together to form peptide bonds.
- There are four types of protein structures: Primary, Secondary, Tertiary, and Quaternary.
- A protein has the amino acid sequence, which is its primary structure.
- Secondary structure is the shape that polypeptide chains form.
- Tertiary structure is the shape that is formed when the secondary structure folds even more.
- Quaternary structure is formed due to the spatial arrangement of sub-units.
FAQs
1. What are the four structures of protein?
There are four types of protein structures: Primary, Secondary, Tertiary, and Quaternary.
2. What is protein structure and function?
When amino acids condense to form peptide bonds, it is known as the protein structure.
We hope you enjoyed studying this lesson and learned something cool about Protein Structures! Join our Discord community to get any questions you may have answered and to engage with other students just like you! We promise it makes studying much more fun! 😎
SOURCES:
- Protein Structure and Levels of Protein: https://byjus.com/chemistry/protein-structure-and-levels-of-protein/. Accessed 28 Feb 2022.
- Protein Structure: https://www.ck12.org/c/chemistry/proteins/lecture/Protein-Structure/.Accessed 28 Feb 2022.