CBSE Class 12 Chemistry Chapter 13 Revision Notes
Chapter 13: Amines Revision Notes
- Amines are ammonia derivatives that have one or more alkyl groups replacing one or more hydrogen atoms.
- Based on alkyl group substitution amines are classified as primary, secondary, or tertiary.
Amino acid preparation
- Nitro compounds are reduced.
- When an alkyl or benzyl halide reacts with an ethanolic solution of ammonia, the halogen atom is replaced by an amino (-NH2) group in a nucleophilic substitution reaction.
- Nitriles are reduced.
- Through amide reduction.
- Synthesis of phthalimide by Ghbriel: It’s only for primary alkyl amines, not aromatic amines.
- Reaction of Hoffmann bromide degradation:
Important amine reactions
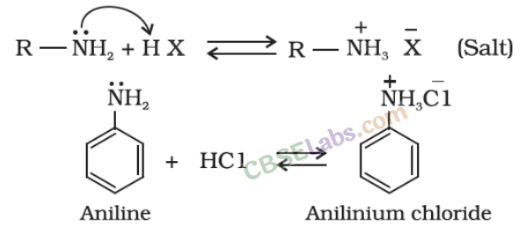
- Amines are basic in nature and form salt when they react with acid.
- Aromatic and aliphatic primary and secondary amines undergo a nucleophilic substitution reaction with acid chlorides, anhydrides, and esters. Acylation is the name for this reaction.
- Aliphatic and aromatic primary amines are converted to isocyanides or carbylamines by heating with chloroform and ethanolic potassium hydroxide. This reaction is not seen to occur in secondary or tertiary amines. The carbylamine reaction, also known as the isocyanide test, is used to detect primary amines.
- Hinsberg’s reagent, also known as CH6H5SO2Cl, reacts with primary and secondary amines to form sulphonamides. Benzene sulphonyl chloride does not react with tertiary amines.
- Diazonium salts have the general formula RN2+ X–, where R denotes an aryl group and X denotes an ion such as Cl”, Br, HSO2–, BF4–, and so on.
- With the evolution of N2, primary aliphatic amines form highly unstable alkyl diazonium salts, which decompose to give alcohols. Primary aromatic amines form arene diazonium salts, which are stable in solution at low temperatures for a short time (273 -278 K).
- The resonance of the arenediazonium ion has been stabilized.
- At 273–278 K, aniline reacts with nitrous acid to produce benzene diazonium chloride. Diazotization is the process of converting primary aromatic amines into diazonium salt.
Chemical Properties
- The diazonium group, which is an excellent leaving group, is substituted by other groups such as Cl–, Br–, I–, CN–, and OH–.
- In the presence of Cu (I) ion, the Cl–, Br–, and CN– nucleophiles can easily be introduced into the benzene ring. This is known as the Sandmeyer reaction.
- The Gatterman reaction is a reaction in which chlorine or bromine is introduced into the benzene ring by treating the diazonium salt solution with the appropriate halogen acid in the presence of copper powder.