Esters Study Guide
INTRODUCTION:
Early in their training, firefighters learn that eliminating one or more legs of the typical “fire triangle” (oxygen, heat, and fuel) will extinguish a fire. They learn to employ various materials (e.g., dry chemical, foam, water) that accomplish one or more of the roles of eliminating heat, oxygen, or fuel from a fire via hose lines and portable fire extinguishers. Saponification (hydrolysis of esters in the presence of alkalis) processes transforming blazing fats to soap, which is non-combustible, in wet chemical fire extinguishers used to put out flames involving fats and oils. Now you must be wondering what an ester is. Let’s talk about that in detail.
DEFINING ESTER
An ester is an organic chemical that is a carboxylic acid derivative in which the hydroxyl group’s hydrogen atom has been substituted with an alkyl group.
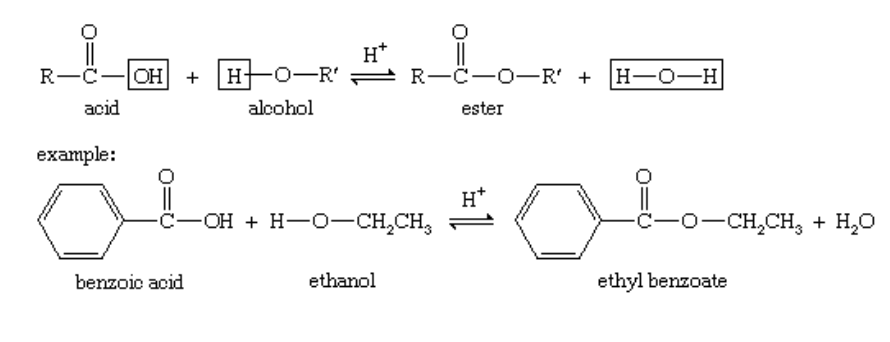
Esters are carboxylic acids that have had the hydroxyl (OH) group replaced with an alkoxy (R-O) group. The condensation of a carboxylic acid with alcohol is a typical way to make them.
A generic ester structure is provided below:
NOMENCLATURE:
Individuals unfamiliar with organic chemistry may be confused by the nomenclature of esters since the name is written in the opposite order as the formula. The ethyl group is listed just before the name in the scenario of ethyl ethanoate, for example. Ethanoic acid is the source of the word “ethanoate.”
While esters’ IUPAC designations are derived from the parent alcohol and acid, many common esters are referred to by their common names. Ethanoate is known as acetate, methanoate is known as formate, propanoate is known as propionate, and butanoate is known as butyrate.
PROPERTIES AND ESTER EXAMPLE:
- Low molecular weight carboxylic acid esters are colorless, effervescent liquids with appealing aromas that are very marginally soluble in water.
- Many are accountable for the aroma and flavor of flowers and fruits; for instance, bananas contain isopentyl acetate, wintergreen has methyl salicylate, and pineapples have ethyl butyrate.
- Artificial flavors, perfumes, and cosmetics include these and other volatile esters with distinct odors.
- Lacquer thinner, paints, and varnishes employ volatile esters as solvents; enormous volumes of ethyl acetate and butyl acetate are commercially manufactured for this reason.
- Animals and plants produce waxes that are esters of long-chain carboxylic acids and long-chain alcohols. Fats and oils are glycerol esters of long-chain carboxylic acids.
CONCLUSION:
- An ester is an organic chemical that is a carboxylic acid derivative in which the hydroxyl group’s hydrogen atom has been substituted with an alkyl group.
- Esters’ IUPAC designations are derived from the parent alcohol and acid.
- Many are accountable for the aroma and flavor of flowers and fruits.
FAQs:
1. What is a common ester?
The most prevalent esters are those made from carboxylic acids.
2. What are ester products?
The scent of numerous fruits, including apples, pears, bananas, pineapples, and strawberries, is due to esters. Artificial flavors, perfumes, and cosmetics include these and other volatile esters with distinct odors.
3. How do you name ester examples?
Determine and name the alkyl group (R’) that has taken the place of the hydroxyl group’s hydrogen. The is the first component of ester. Replace the word’s ending from –oic acid to –oate for the carboxylic acid part of the molecule (R-COO). The second component of the ester name would be this.
We hope you enjoyed studying this lesson and learned something cool about Esters! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun, VR classrooms – we promise, it makes studying much more fun! 😎
SOURCES:
- Ester general formula. https://www.ck12.org/c/chemistry/esters/lesson/Esters-CHEM/. Accessed 26 Jan 2022.
- Nomenclature. https://www.thoughtco.com/definition-of-ester-605106. Accessed 26 Jan 2022.
- Ester. https://www.britannica.com/science/ester-chemical-compound. Accessed 26 Jan 2022.