Phase Diagram for Water Study Guide
INTRODUCTION
Before understanding the phase diagram of water, one needs to understand the basics: what is a phase diagram? A phase diagram is a graphical representation of the phase change of different substances under different pressure and temperature conditions. In physical chemistry, mineralogy, material science, and engineering, the phase diagram is a chart that showcases the conditions at which thermodynamically distant phases occur as well as coexist at equilibrium.
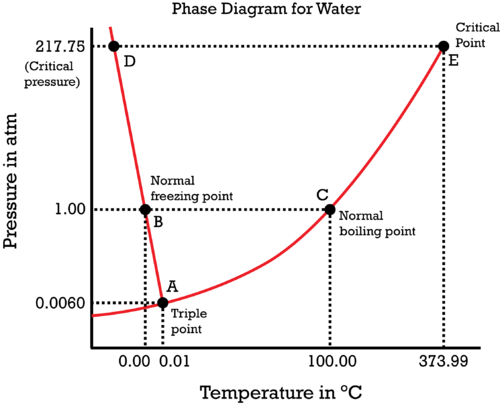
What is the phase diagram of water? It is a pressure-temperature diagram that showcases water in its various states like ice, liquid, and steam under different conditions.
In this diagram, the X-axis and Y-axis represent temperature and pressure, respectively. The colors blue, green, and yellow represent the different water states. As you can observe, the first curve becomes the boundary between the gas and the solid phase, while the second phase becomes the boundary between the liquid and the solid phase. The third curve then becomes the boundary between the gas and the liquid phase, and this curve is also regarded as the saturated line.
The water phase you will observe at this line is the saturated steam, and along the saturated line, no matter the pressure, temperature condition, and magnitude, water and steam would be present in equilibrium. The triple point of water is the end of the first curve, and at this point, the three phases of water- solid, liquid, and steam coexist. The pressure of this particular condition is 0.61 kPa and 0.010 degrees Celsius.
As you can observe from the diagram, just below the triple point, water does not have any liquid phase. Under the triple point of water, with temperature rise, ice would instantly become vapor at constant pressure. We can again classify the steam phase into three distinctive phases- saturated steam, superheated steam, and supercritical steam.
Above the triple point in the phases of the water diagram, we observe two branching curves with each function. One curve is between solid and liquid, while the other can be observed between gas and liquid phases. The critical point is present at the end of the gas-liquid curve. The critical point of water is at 22.1 MPa and 374 degrees Celsius.
Water vapor at temperature and pressure right above the critical point cannot be condensed just by increasing the pressure. If you heat water at a constant pressure of 25 MPa, the liquid phase of water will not experience a saturated steam phase but would directly turn into supercritical vapors. This concept is primarily used in steam power plants.
CONCLUSION:
- The water phase diagram is a pressure-temperature diagram that represents various water phases, including water, steam, and ice, under different conditions.
- The phase diagram is known to include phase boundaries.
FAQs:
1. What are the three parts of a phase diagram?
The three parts of a phase diagram are liquid, gaseous, and solid states.
2. What is a phase diagram of a water system?
It is a pressure-temperature diagram of water in its various states like ice, liquid, and steam under different conditions.
We hope you enjoyed studying this lesson and learned something cool about the Phases Of Water Diagram! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our app to experience our fun VR classrooms – we promise it makes studying much more fun! 😎
SOURCES:
- Phase Diagram of Water: https://medium.com/@ashwinpalo/phase-diagram-of-water-definition-and-explanation-4ca37a553033. Accessed 28 Feb 2022.
- Phase Diagram of Water: https://courses.lumenlearning.com/cheminter/chapter/phase-diagram-for-water/. Accessed 28 Feb 2022.
- Phase Diagram of Water: https://www.ck12.org/c/chemistry/phase-diagram-for-water/rwa/All-Three-Phases-AT-Once/ Accessed 28 Feb 2022.