Spontaneous And Non-Spontaneous Reactions Study Guide
Introduction
Take an ice cube out of the refrigerator and put it in a bowl at room temperature. What happens? It melts. But the important thing here is that we had no interference during that melting process. It just happened on its own. This is a typical example of a spontaneous reaction. So, wondering what a spontaneous reaction is? Let’s find out.
What Is A Spontaneous Process?
- The term “spontaneous reaction” refers to a reaction that promotes the creation of products under the circumstances that it occurs.
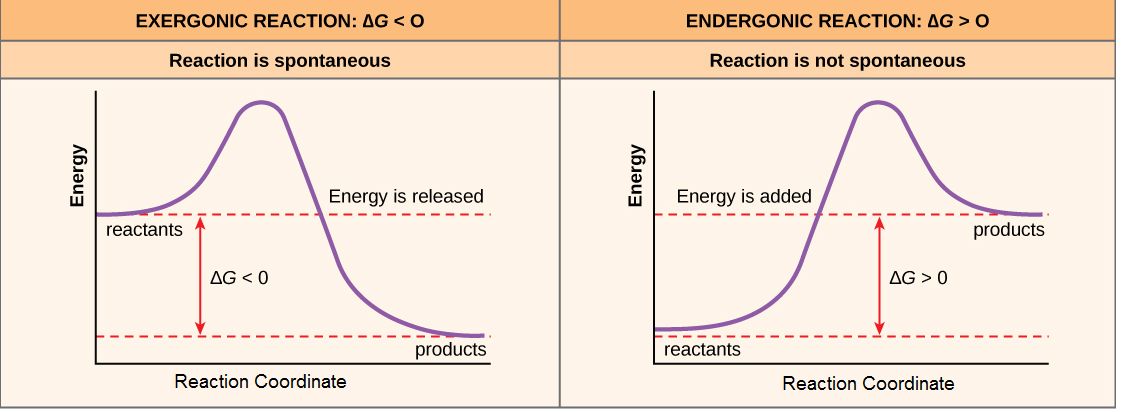
- A spontaneous process can move in a specific direction without an external energy source.
- The rules of thermodynamics (specifically the 2nd law of thermodynamics) determine the direction of a spontaneous process, assuring that if a significant number of specific interactions are engaged, the path will always be toward rising entropy.
The Second Law Of Thermodynamics
-
The second rule of thermodynamics dictates that the total (change in entropy S) ΔS must be larger than or equal to 0 for every spontaneous process; however, spontaneous chemical changes could lead to a negative change in entropy.
-
However, because such a process must have a relatively substantial negative change in enthalpy, this does not defy the second rule (heat energy).
-
The rise in temperature of the reaction environment causes a significant increase in entropy, resulting in a positive net change in entropy.
-
That is, the surroundings’ (change in entropy S) ΔS increases sufficiently as a result of the reaction’s exothermicity (release of heat during a chemical reaction) to counterbalance for the system’s negative (change in entropy S) ΔS.
Note – A substance’s entropy (S) is a thermodynamic property that is directly proportional to its degree of disorder. The more possible microstates (arrangement of molecules) for a system, the higher the disorder and the greater the entropy.
Spontaneous Process:
Spontaneity does not entail that the reaction happens quickly. The pace of a reaction is determined by the chemical kinetics of the reaction rather than its spontaneity. In a spontaneous process, every reactant tends to generate the corresponding product. This pattern is linked to stability.
Non-Spontaneous Process:
- A chemical reaction known as an endergonic reaction (also known as a nonspontaneous reaction) is one in which the standard change in free energy is positive, and energy is absorbed. Because the whole quantity of energy represents a loss, the net outcome is negative.
- Endergonic processes can also be driven by connecting them through a common intermediate to a highly exergonic reaction.
For example: PhotosynthesisDuring photosynthesis, plants use energy from the sun to turn carbon dioxide (CO2) and water (H2O) into sugar (C6H12O6) and oxygen (O2).6 CO2 (g) + 6 H2O (l) + energy → C6H12O6 (s) + 6 O2 (g)
Summary
- A spontaneous reaction happens in a given set of conditions without any interruptions.
- The nonspontaneous reaction takes place with the help of external agents like heat or energy.
FAQs:
1. What is a spontaneous and nonspontaneous reaction?
A spontaneous reaction takes place without any external agent. An example is the melting of ice. This doesn’t require any external agent. Happens slowly and steadily. A non-spontaneous reaction requires an external agent like heat or light or energy of some sort to make the reaction start. For example, photosynthesis. A plant absorbs water and carbon dioxide. But for it to get converted, sunlight is required. This is a nonspontaneous reaction.
2. How do you know if a reaction is spontaneous or nonspontaneous?
If the reaction takes place without any external agent, it’s a spontaneous reaction. If the reaction takes place with the help of an external agent, then it’s a nonspontaneous reaction.
We hope you enjoyed studying this lesson and learned something cool about Spontaneous and non-spontaneous reactions! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun, VR classrooms – we promise, it makes studying much more fun!😎
Sources:
- Spontaneous and Nonspontaneous reaction. https://www.ck12.org/c/chemistry/spontaneous-and-nonspontaneous-reactions/lesson/Spontaneous-and-Nonspontaneous-Reactions-CHEM/. Accessed on 26th Jan 2022.
- Spontaneous and Nonspontaneous reaction. https://www.chemistrylearner.com/chemical-reactions/spontaneous-and-non-spontaneous-reaction. Accessed on 26th Jan 2022.
- Spontaneity https://byjus.com/chemistry/spontaneity/, Accessed on 26th Jan 2022.