Different Types of Mixtures Study Guide
INTRODUCTION
When you get up in the morning, you first wash your face with water and brush your teeth. Toothpaste is a mixture of different materials which help to clean your teeth. Again when you are having your food, the food is prepared with a different mixture of spices and different vegetables all mixed together. A mixture is known to be the blend of two or more substances together.
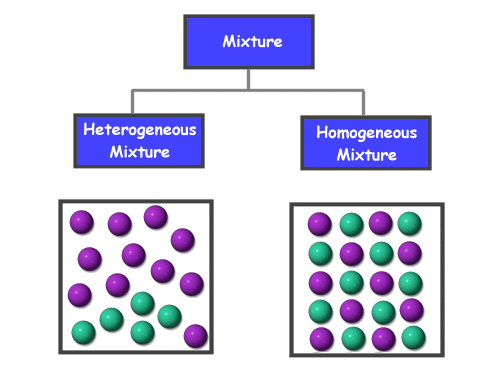
Now, the mixture can be divided into two parts:
- Homogenous mixture
- Heterogeneous mixture
Let us know more about these mixtures!
HOMOGENOUS MIXTURE
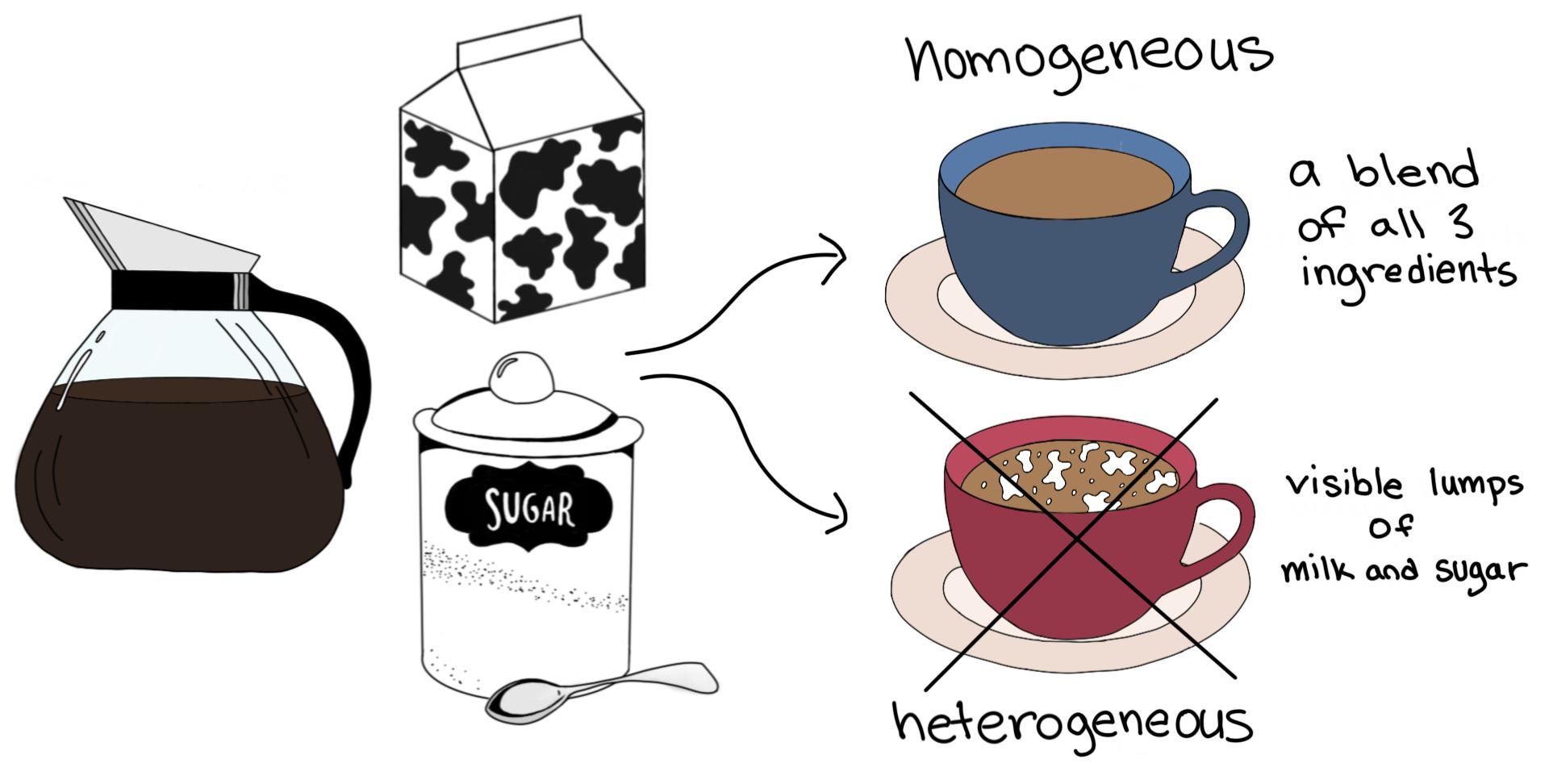
A homogeneous mixture can be defined as a uniformly distributed mixture. All the components present in the homogenous mixture are mixed together consistently so that the proportions are of the same ratios. We can observe only one phase of matter at one time in a homogenous mixture. Hence, we will not observe solid and gas or liquid and gas, etc. Examples of homogeneous mixtures: air, seawater, blood, any juice, etc.
HETEROGENEOUS MIXTURE
Heterogeneous mixtures can be defined as non-uniform mixtures of different components. All the components present can be distinguished and identical to each other. All the components present have localized regions with various distinct properties. We will find more than two phases present, and we can identify them based on their characteristic properties. We can even distinguish the phases even if they are of the same matter.
Examples of heterogeneous mixtures: oil and water, vegetable soup, mixed nuts, etc.
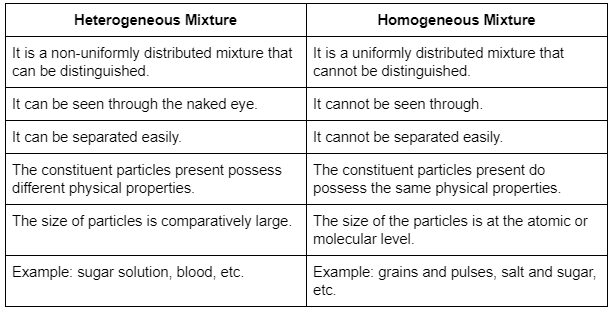
DIFFERENCE BETWEEN HOMOGENEOUS AND HETEROGENEOUS MIXTURES
Differences between homogeneous and heterogeneous are as follows:
FAQs
1. What is a homogeneous mixture? Give an example.
A homogeneous mixture is a type of mixture in which the components are mixed together in equal proportion. An example of a homogeneous mixture is saline solution, different alloys, etc.
2. What are the 5 examples of a homogeneous mixture?
Five examples of homogeneous mixtures are as follows:
- Air
- Wine
- Seawater
- Blood
- Steel
3. Write two differences between homogeneous and heterogeneous mixtures.

We hope you enjoyed studying this lesson and learned something cool about Homogeneous Vs. Heterogeneous Mixture! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our app to experience our fun VR classrooms – we promise it makes studying much more fun! 😎
SOURCES
- Heterogeneous and Homogeneous Mixtures: https://www.thoughtco.com/heterogeneous-and-homogeneous-mixtures-606106. Accessed 5th March 2022.
- Types of Mixtures: https://www.khanacademy.org/science/ap-chemistry-beta/x2eef969c74e0d802:iintermolecular-forces-and-properties/x2eef969c74e0d802:solutions-and-mixtures/v/types-of-mixtures#:~:text=There%20are%20two%20types%20of,solid%2C%20liquid%2C%20or%20gas.Accessed 5th March 2022.