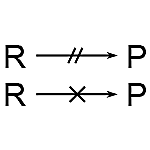Signs of Chemical Reactions Study Guide
INTRODUCTION:
Have you ever wondered what an arrow signifies in a chemical reaction? Why is there an arrow separating two different groups of chemicals like CH₄(g) + 2O₂(g)→CO₂(g) + 2H₂O(g)?
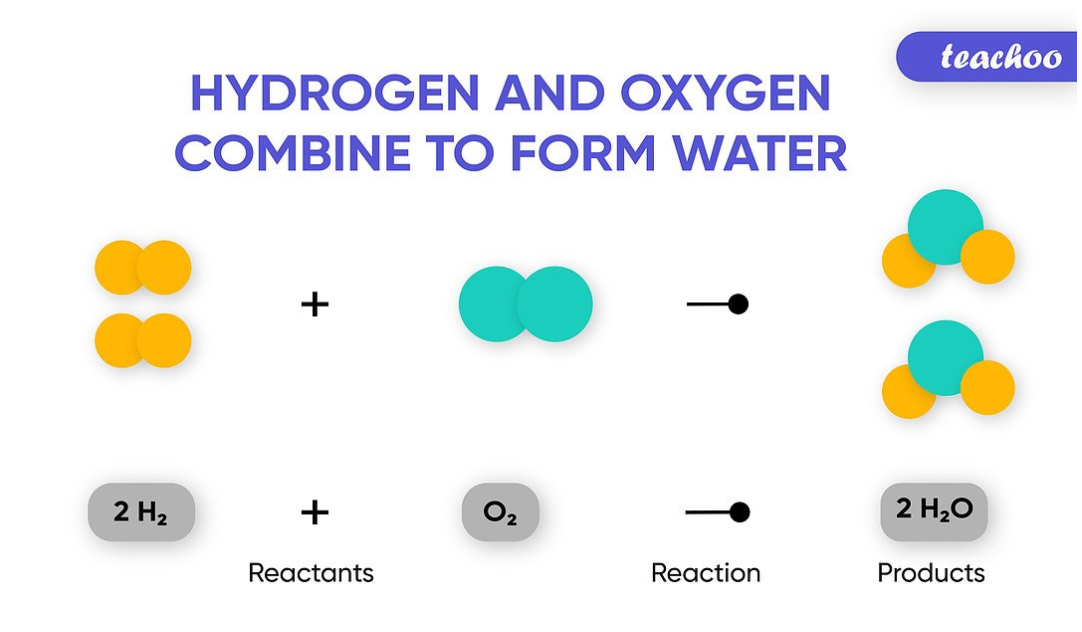
This is a chemical equation. The symbolic representation of a chemical change is a chemical equation. The reactants (the chemicals that initiate the chemical reaction) are represented on the left, while the products (the substances that result from the reaction) are represented on the right. An arrow sign (“→” commonly read aloud as “yields”) separates the two.
The state attributes of products and reactants, either aqueous (dissolved in water — aq), solid (s), liquid (l), or gas, should be included in chemical equations.
USE OF ARROW IN CHEMICAL EQUATIONS
When a reaction necessitates energy use, it is frequently shown above the arrow. If energy is provided to the reaction in the form of heat, a capital Greek letter delta (Δ) is placed on top of the reaction arrow; if energy is supplied in the form of light, hv is inscribed.
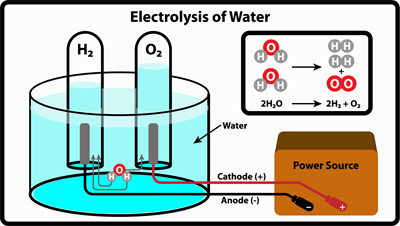
There are a wide variety of reactions possible in our everyday lives: Elements can form compounds (as shown in the reaction), compounds can generate elements (water will split in the presence of a current flow to release hydrogen gas and oxygen gas), or compounds can mix, split away, or reorganize to form new compounds.
TYPES OF ARROWS IN A REACTION:
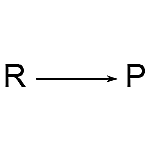
1. Right Arrow: The right arrow is the most prevalent arrow in a chemical reaction formula. The direction indicates the reaction’s direction.
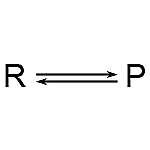
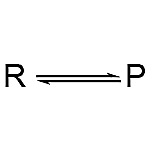
2. Double Arrow: A reversible reaction is indicated by the double arrow.
3. Equilibrium Arrow: When the reaction is at equilibrium, two arrows with solitary spikes directed in opposing directions suggest a reversible reaction.
4. Broken or Crossed Arrow: An arrow with a centered double hash or cross indicates that a response cannot occur. Broken arrows can also indicate reactions that were attempted but failed.
CONCLUSION:
- A chemical equation is an empirical representation of a chemical process.
- Base materials are represented on the left-hand side of the equation as reactants.
- The right-hand side of the equation contains the products, which are the reaction results.
- A chemical or physical transition is represented by the arrow.
- In chemical reaction formulae, the right arrow is by far the most prevalent arrow. The direction indicates the reaction’s direction.
FAQs:
1. What is an arrow in a chemical equation?
Chemical equations are written in a standard format. The reactants are listed on the left side of the equation, while the products are listed on the right. The direction of the reaction is shown by an arrow pointing from the reactants to the products.
2. What does a crossed arrow mean in chemistry?
An arrow with a centered double hash or cross indicates that a response cannot occur. Broken arrows can also be used to indicate reactions that were attempted but failed.
We hope you enjoyed studying this lesson and learned something cool about What does an arrow mean in a chemical equation! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun, VR classrooms – we promise, it makes studying much more fun! 😎
Continue your learning journey
Learn about the States of matter
Learn about the Structure of water
SOURCES:
- Reactants and Products. https://www.ck12.org/c/chemistry/reactants-and-products/lesson/Reactants-and-Products-CHEM/. Accessed 24 Jan 2022.
- Experimental Data and Empirical Formulas. https://courses.lumenlearning.com/boundless-chemistry/chapter/experimental-data-and-empirical-formulas/. Accessed 24 Jan 2022.
- What do you read the arrow in a chemical equation as meaning?. https://socratic.org/questions/what-do-you-read-the-arrow-in-a-chemical-equation-as-meaning. Accessed 24 Jan 2022.