CBSE Class 10 Science Chapter 5 Revision Notes
Chapter 5: Periodic Classification of Elements Revision Notes
Dobereiner’s Triads
- Dobereiner named a trio after arranging a group of three elements with comparable characteristics in order of increasing atomic masses.
- He demonstrated that the middle element’s atomic mass is about equal to the arithmetic mean of the other two.
- However, Dobereiner was only able to determine the following three triads using the materials available at the time.
Newlands’ Law of Octaves
- The physical and chemical characteristics of every eighth element are comparable to those of the first when the elements are grouped according to increasing atomic masses.
- These octaves, according to Newlands, are similar to the eight notes of a musical scale.
Limitations and Assumptions:
-
The law applied to atoms having atomic masses between 40 and 100.
-
The properties of newly found elements did not conform to the law of octaves.
-
To accommodate components in the table, Newlands put two elements in the same position in a few situations.
-
He also put parts that were dissimilar into the same slot.
The Periodic Table and Law of Mendeleev
- Elements’ physical and chemical characteristics follow a regular pattern based on their atomic weights.
Mendeleev’s Periodic Table Features
- Periods are made up of twelve horizontal rows that have been shortened to seven.
- Groups are a set of eight vertical columns.
- A and B subgroups were created from Groups I through VII.
- There are no subgroups in Group VIII, and each row has three items.
- The characteristics of elements in the same group are comparable.
Mendeleev’s Periodic Table’s Achievements
-
An organised study of elements: Elements having comparable chemical and physical characteristics were grouped together to make studying their chemical and physical properties easier.
-
Atomic mass correction: The placement of atoms in Mendeleev’s periodic table aided in the correction of some elements’ atomic masses. The atomic mass of beryllium, for example, was changed from 13.5 to 9. Indium, gold, platinum, and other atomic masses were also rectified.
-
Prediction of attributes of yet-to-be-discovered elements: Yet-to-be-discovered elements were given the names eka-boron, eka-aluminium, and eka-silicon. The characteristics of these elements might be correctly predicted based on the properties of other elements in the same group. These elements were given the names scandium, gallium, and germanium when they were found.
-
Noble gases were found and readily placed in a new group known as the zero group of Mendeleev’s table, without disrupting the previous arrangement.
Mendeleev’s Periodic Table’s Limitations
-
Hydrogen’s place: Because hydrogen has qualities with both alkali metals (IA) and halogens (VIIA), Mendeleev was unable to defend its location.
-
Isotope Position: Isotopes have various atomic weights, yet they are not arranged in distinct locations in Mendeleev’s periodic table.
-
Cobalt (Co) has a larger atomic weight than Nickel (Ni), however it was put before Nickel (Ni) on the periodic table.
-
Comparable elements are classified in distinct groups: Platinum (Pt) and Gold (Au) have similar qualities, yet they are placed in separate groups.
-
Periodicity’s Cause: He was stumped as to what caused periodicity among the elements.
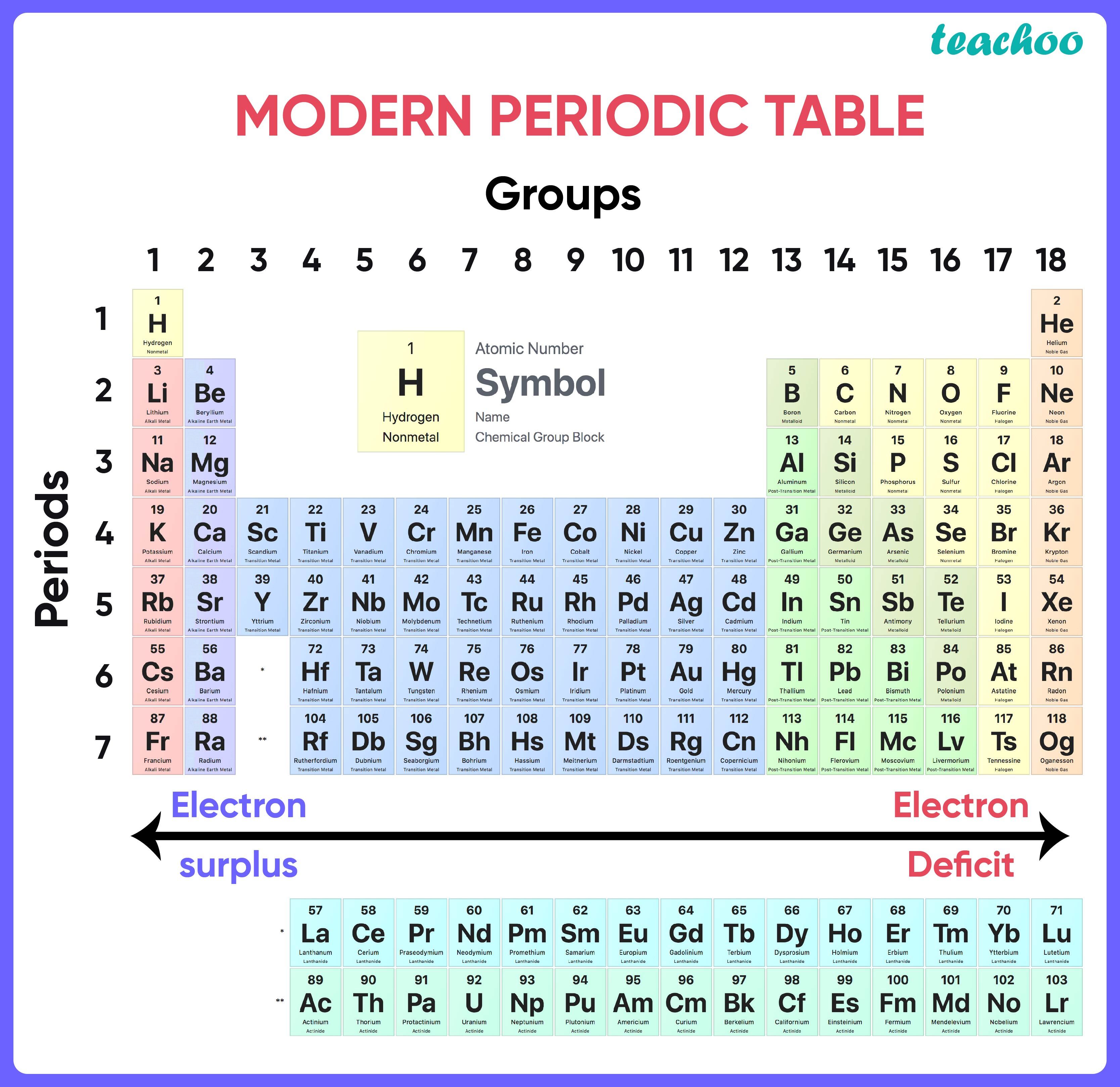
MODERN PERIODIC TABLE
- The periodic function of an element’s atomic number determines its physical and chemical characteristics.
- The repeating of the same outer shell electrical structure at a regular interval is the cause of periodicity.
Periods in the Periodic Table of the Modern Era
The number of shells present in elements from the same era is equal to the period number.
The number of electrons in the valence shell grows from one to eight as you move from left to right in a given time, but the number of shells stays the same.
Number of Elements in a Period
- There are just two components in the first period. The shortest duration is known as 1H and 2He.
- Short periods are the second (3Li to 10Ne) and third (11Na to 18Ar) periods, which each include eight elements.
- Long periods are the fourth (19K to 36Kr) and fifth (37Rb to 54Xe) periods, which each include 18 components.
- The sixth period, generally known as the longest period, includes 32 components (55Cs and 86Rn).
- The seventh quarter is a gap in the game.
- (The seventh period is now officially complete, thanks to recent discoveries of new elements and their insertion to the periodic table.)
Groups in Modern Periodic Table
- Groups are the 18 vertical columns that make up the contemporary periodic table.
- Alkali metals belong to Group 1 of the periodic table.
- Alkaline earth metals are the second group of elements.
- Pnicogens are elements that belong to Group 15.
- Chalcogens are elements that belong to Group 16.
- Halogens are elements in Group 17 of the periodic table.
- Noble gases are elements in Group 18 of the periodic table.
Alkali Metals
Alkali metals are the elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr) in the first group.
Because they all react with water to generate alkalis, they were given that name.
At normal temperature and pressure, the alkali metals are all lustrous, soft, and highly reactive solids that rapidly shed their outermost electron to create cations with charge +1.
The number of valence electrons is equal to one.
Alkali Earth Metals are metals that have a high alkali content.
Alkaline earth metals
are the elements beryllium (Be), magnesium (Mg), calcium (Ca), strontium (Sr), barium (Ba), and radium (Ra) in the second group.
Their name comes from the fact that their oxides are naturally alkaline.
At normal temperature and pressure, they are all lustrous, silvery-white, moderately reactive hard solids. To produce cations with a charge of +2, they lose two electrons from their outermost shell.
The number of valence electrons is equal to two.
Halogens
- The sixteenth group of elements (F, Cl, Br, I, and As) are known as halogens and exist as diatomic molecules.
- The letter ‘X’ is frequently used to refer to any halogen.
- Because they all generate a wide spectrum of salts when interacting with metals, they were given the term halogen, which comes from the Greek words Hal (“salt”) and gen (“to produce”).
- At room temperature, halogens exist in all three states of matter: Iodine and astatine are solids. Bromine in liquid form. Fluorine and chlorine are two gases.
- The number of valence electrons in a molecule is 7.
Noble Gases
- Helium (He), neon (Ne), argon (Ar), krypton (Kr), xenon (Xe), and the radioactive radon (Rn) belong to the eighteenth group of elements.
- They’re all odourless, colourless, monatomic gases with negligible chemical reactivity.
- They have limited inclination to engage in chemical processes since their valence shell is deemed “full.”
- Scientists thought these gases were extremely uncommon and chemically inert when they were found and classified, therefore they were given the titles ‘rare’ and ‘inert’ gases.
- The number of valence electrons is equal to eight.
CLASSIFICATION OF THE MODERN PERIODIC TABLE
- Metals, non-metals, and metalloids are all categories in the contemporary periodic table.
Metals
- Metals are electropositive because they lose electrons to form bonds.
- Metal oxides are, in most situations, basic in nature.
Non-metals
- Nonmetals are electronegative because they gain electrons to form bonds.
- Non-metal oxides are acidic in nature in most circumstances.
Metalloids
- Metalloids and semimetals are elements that have characteristics of both metals and nonmetals.
- Boron, silicon, germanium, arsenic, antimony, tellurium, and polonium are just a few examples.
TRENDS IN THE MODERN PERIODIC TABLE
- Elements in a group have the same number of valence electrons.
- The quantity of shells grows as you progress through the group.
- Elements in a period have the same number of shells.
- The number of valence shell electrons increases by one unit throughout the time.
Variation of Valency
- The electrical configuration may be used to compute the value of an element in two ways:
- Number of valence electrons = valency (if they are 1, 2, 3 or 4).
- The number of valence electrons is equal to the number of valence electrons (if they are 5, 6, 7 or 8).
- A group’s members all have the same number of valence electrons. As a result, they have the same valency.
Variation of Atomic Size
- Atomic size, also known as radii, is the distance between the nucleus and the valence shell of an atom.
- The atomic radius reduces over time as the effective nuclear charge increases by one unit, attracting valence electrons or the electron cloud to the nucleus.
- Down the group — As new shells are added, the distance between the nucleus and the valence electrons or electron cloud grows.
Variation of Metallic Properties
- Metallic character reduces with time as the tendency to lose valence electrons decreases as nuclear charge increases.
- Nuclear pull reduces as the distance between the nucleus and the outermost electron grows. This enhances an atom’s ability to lose valence electrons, resulting in an increase in metallic character.
Variation of Nonmetallic Properties
- As the ability to acquire electrons in the valence shell grows due to increased nuclear charge, non-metallic nature increases.
- Nuclear pull reduces as the distance between the nucleus and the valence shell rises.
- As a result, an atom’s potential to gain an electron in its valence shell reduces, and its non-metallic nature decreases.
Variation of Electronegativity
- Electronegativity grows with time as the potential to acquire electrons in the valence shell increases as nuclear charge increases.
- Nuclear pull reduces as the distance between the nucleus and the valence shell rises.
- This reduces an atom’s potential to acquire an electron, lowering electronegativity.