Reaction Order Study Guide
INTRODUCTION
Reactions such as rusting take a lot of time to occur, while a neutralization reaction, i.e., when an acid reacts with a base, takes almost no time. So how fast or slow a reaction is, is called the rate of reaction, and the rate of reaction depends on the order. You must be wondering what reaction order is in chemistry? Let’s understand.
REACTION ORDER
Order of reaction is the sum of the power of concentration of reactants written in rate law expression. Let’s understand in simple words.The rate of a reaction depends or sometimes doesn’t depend on the concentration of reactants.
Reaction rate = is the change in the amount of a reactant or product per unit time.
So, the reaction order is the relationship between the concentrations of species and the rate of a reaction. The order of reaction can be any integer or fraction. It can be found out only experimentally.
How to Determine Reaction Order
The sequence of the reactions may be ascertained using a number of different techniques. This part describes a few of these techniques.
Initial Rates Method
First, the power-law expression’s natural logarithm form is derived. It is determined by the formula: ln r = ln k + x.ln[A] + y.ln[B] +….
Now, the reaction is run with changing concentrations of the reactant in issue while maintaining a constant concentration for the other reactants to compute the partial order corresponding to each reactant.
The power-law version of the rate equation is now ln r = x.ln[A] + C, where C is a constant if the partial order of A is being determined.Now, a graph is drawn using ‘ln r’ as a function of ln[A], with the partial order, denoted by x, as the appropriate slope.
Integral Method
This approach is often used to confirm the reaction order determined using the initial rates method.The integral version of the rate law is used to compare the reactant concentrations that have been measured.
For instance, if the value of ln[A] corresponds to a linear function of time (integrated rate equation of a first-order reaction: ln[A] = -kt + ln[A]0), the rate law for the first-order reaction is confirmed.
Differential Method
The simplest approach for determining the sequence of reactions is this one.First, the reaction’s rate expression (r = k[A]x[B]y.) is written.The ultimate value of the reaction order is determined by adding the exponents x+y+…
In addition to these techniques, there are other approaches to determining the order of a reaction, such as the flooding technique, which measures the concentration of a single reactant while all the other reactants are present in massive excess.
DIFFERENT RATE ORDERS
ZERO-ORDER REACTIONS
A → BHere, the rate is independent of the concentration of the reactant, which is expressed as [A].
Rate = k[A]0The power of [A] is 0, and hence the order of the reaction is zero. This means the rate of reaction won’t change even if we increase or decrease the concentration of the reactant.
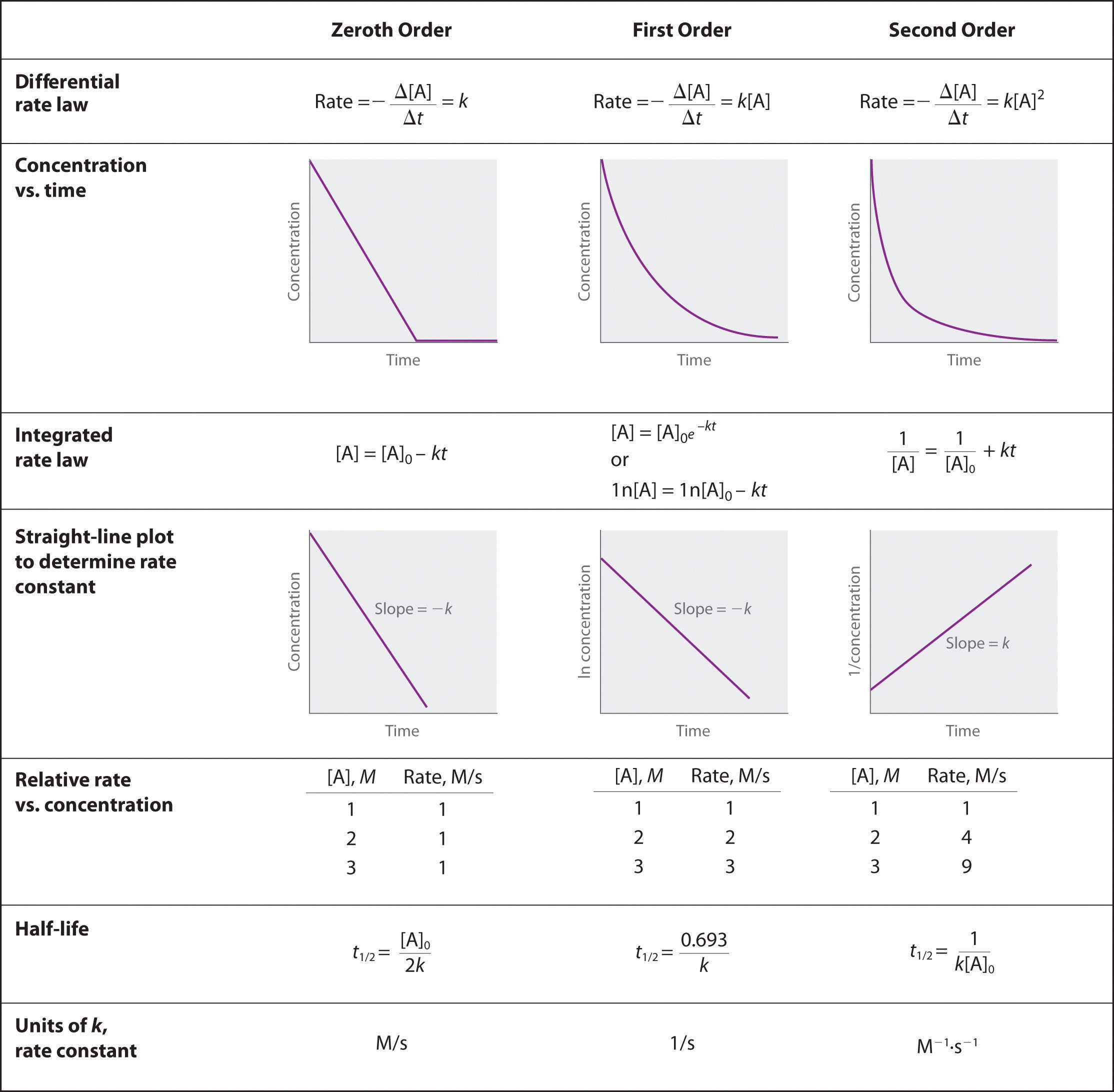
FIRST-ORDER REACTIONS
A → BHere, the rate of reaction is proportional to the concentration of A. This means as [A] increases, the rate of reaction increases too and vice versa.
Rate = k [A]Since the power of [A] is 1, the order of the reaction is 1. The rate depends linearly on the concentration of reactant here. The rate of reaction is the slope of the graph.
SECOND-ORDER REACTION
A + B → CHere, the rate is expressed as k [A] [B], which means the rate will be dependent on both of the reactants A and B. If we increase or decrease the concentration of any of these, then the rate will change.
The rate is proportional to [A], and the order for A is 1, and the same is for B. But the overall order is the sum of orders with respect to both the reactants, so it will be 2.
Difference Between Molecularity and Order of Reaction
| Molecularity | Order of Reaction |
|---|---|
| It is the number of molecules participating in the step that determines the rate. | It illustrates the connection between reactant concentration and reaction rate. |
| Only the rate-assessing step is taken into account for determining molecularity. | All stages of a reaction are taken into account while determining the order of the reaction. |
| It doesn’t depend on pressure and temperature. | It depends on pressure, temperature and concentration. |
CONCLUSION
- Order of reaction is the sum of the power of concentration of reactants written in rate law expression.
- It can be an integer or fraction.
FAQs
1. What is the order of a reaction?
Order of reaction is the sum of the power of concentration of reactants written in rate law expression.
2. Can the order of reaction be -½?
Yes, the order of the reaction can be an integer or fraction.
3. For Rate = k [A], what is the order of reaction?
Since the power of concentration of reactant is 1, the reaction is of the first order.
We hope you enjoyed studying this lesson and learned something cool about Reaction Order! Join our Discord community to get any questions you may have answered and to engage with other students just like you! Don’t forget to download our App to experience our fun VR classrooms – we promise it makes studying much more fun! 😎
]]>